

Drug Facts |
Health & Safety |
Usage Instructions |
About this Product |
Company, Brand & Sustainability |
|
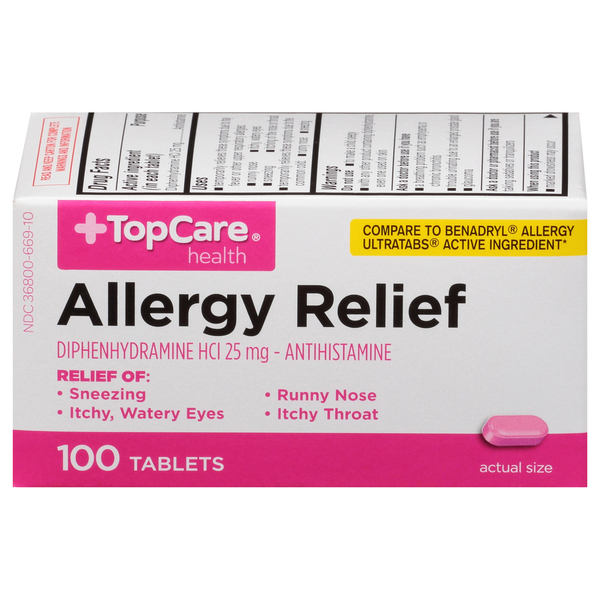
Adults and Children 12 Years and Over
|
1 to 2 tablets.
|
|
Children 6 to Under 12 Years
|
1 tablet.
|
|
Children Under 6 Years
|
Do not use.
|
Product Allergens
Expiration Date
This product has an expiration date. Refer to expiration date stamped on packaging.
Ask a doctor or pharmacist before use if you are taking sedatives or tranquilizers.
If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222).
Tamper Evident: Do not use if safety seal under cap is broken or missing.
About this Product
Certifications
SmartLabel® includes certifications from independent organizations that have meaningful and consistent standards for product composition, environmental protection and/or social justice.
Expiration Date
This product has an expiration date. Refer to expiration date stamped on packaging.
Country of Origin Statement
Product Allergens
ACTIVE INGREDIENTS
INACTIVE INGREDIENTS
Adults and Children 12 Years and Over
Children 6 to Under 12 Years
Children Under 6 Years